Today, I took my science. It is a new class because a year has passed. Today, we learned about atoms, Elements, and Ions. I will explain about them.
First, we need to know what an atom is. They are the smallest thing that makes an object. They are divided into molecules. There are more than a hundred atoms, and there is a chart for this. A periodic table!

https://t1.daumcdn.net/cfile/tistory/9958EE445C8CF78621
So, to name some, H, He, U, Be, B, C, N, O, F, Ne, Na, Mg… Today, we needed to memorize this until number 20. (Of course, in Korean) It was very hard. Anyways, I will continue explaining about atoms. So, the things in the periodic table are all atoms. We think that there are many things that makeup objects, but there are only 118! So small than we expected, right? Now, I will explain the history.
At earlier times, this Roman person thought that 4 things make up everything – wood, fire, water, land. However, as times passed people found out that it was wrong. Elements were just elements. There are atoms, so people made the periodic table. Now, I will tell you the things that are inside the atom.
To start with, I want you to look at this picture.
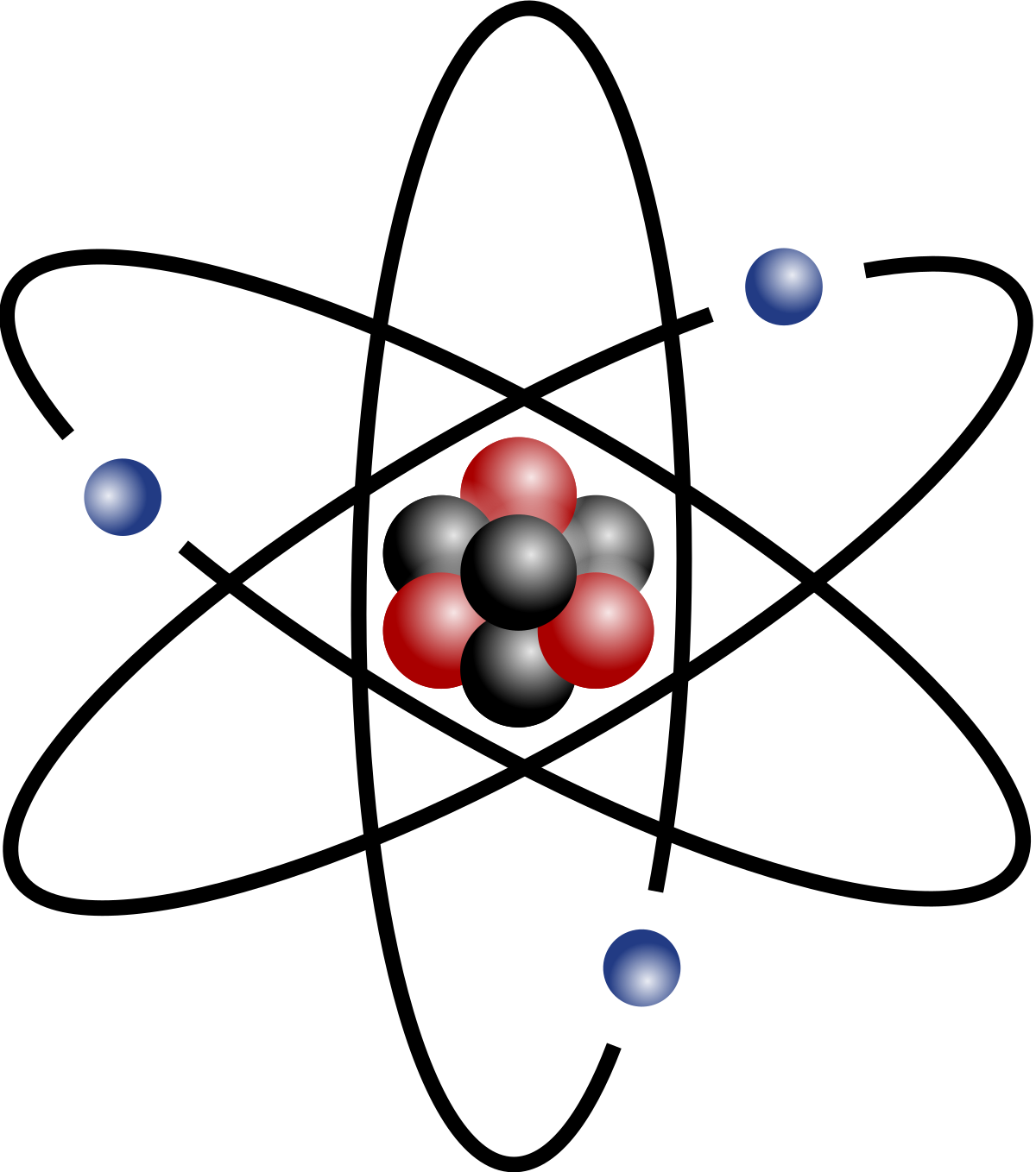
https://ko.wikipedia.org/wiki/%EB%9F%AC%EB%8D%94%ED%8D%BC%EB%93%9C_%EC%9B%90%EC%9E%90_%EB%AA%A8%ED%98%95
The thing in the middle is called an atomic nucleus. It consists of protons and neutrons. The blue thing that is rotating the atomic nucleus is the former. The number of protons and formers is the same. It depends on the number in the periodic table. For example, let’s look at H. It is number one, meaning that there is only one proton and one former. For the neutrons, It is mostly the same. Now, how do we draw an atom?
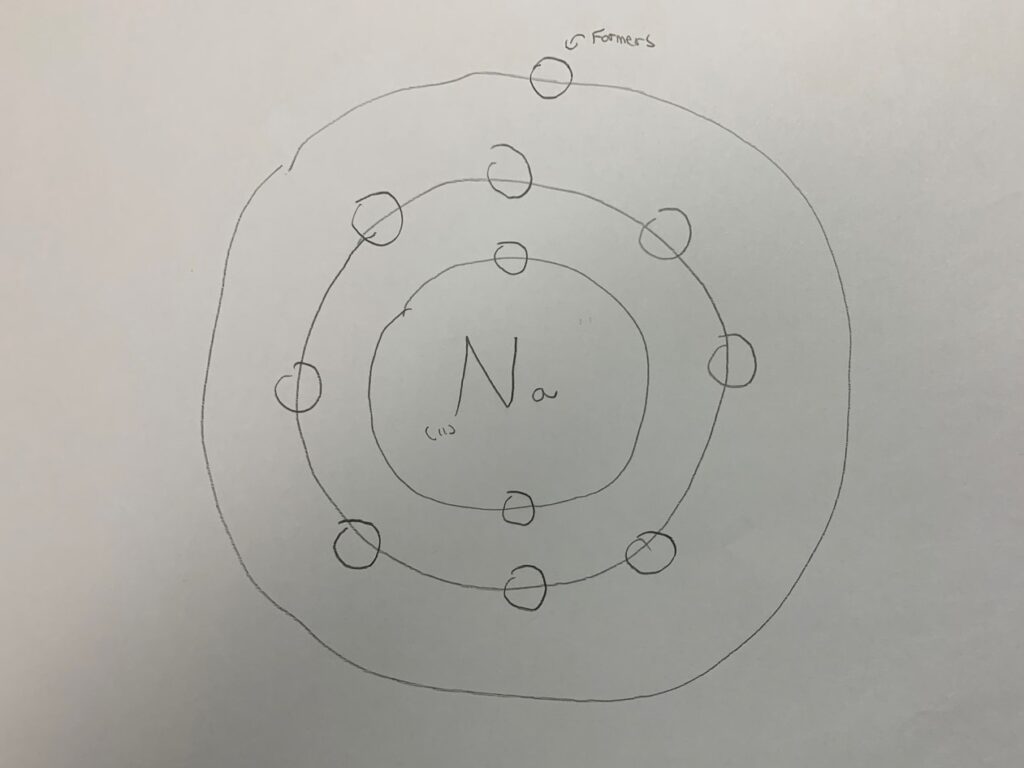
I drew this. You can see 3 circles around Na. On the very inner circle, there are 2 circles, the middle circle consists of 8 circles, and the last circle consists of 1 circle. That is because Na is number 11. 2+8+1= 11. There is a rule in drawing an atom. The very inside circle consists of only 2 circles, the middle one eight, and the last one can contain up to 18. Now, I will explain about positive ions and negative ions.
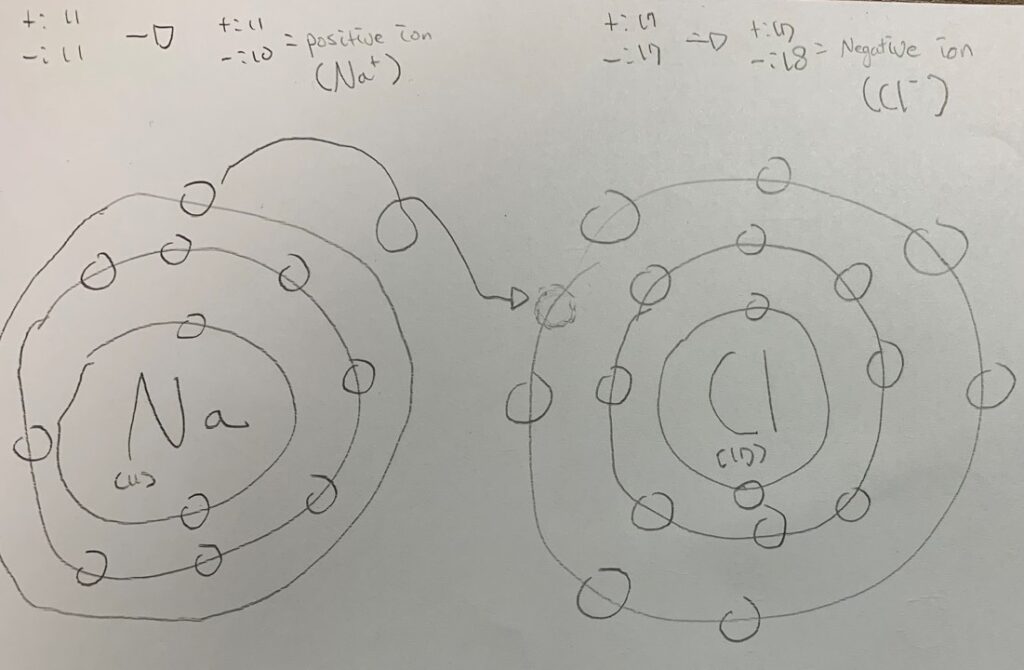
So, remember that I explained about the circles and the number of formers? Well, I drew two atoms which are Na(11) and Cl(17). Most of the atoms want their very outer layer’s former’s number to be eight. However, Na has one, and if we get rid of that one former, there are 8 at the very outer layer. However, Cl has 7 on the outer layer, so we need to get one. One wants to give, while the other wants to get. They fit together. This makes NaCl (you probably heard of this) At first, Na has 11 pluses and 11 minutes. However, as Na gave one, it is a positive ion because the positive one is greater than the negative one. Opposite with Cl.
So, this was what I learned at my science academy. Bye!
Ohmygod
Whydoyoustudysohard stuff daniel
Im really bad at science lel
Ohmygod
Whydoyoustudysohard stuff daniel
Im really bad at science lelll